- Blog
- Will modbus poll work with ab plcs
- Basic lettering lessons tajima pulse
- Nikon scan 4000 ed bit depth
- Winchester model 25 hornet magazine
- Free download parasite in city android
- Harley davidson golf cart parts 50252-74
- Eternal poison cheat engine
- Quantum harmonic oscillator
- Install haproxy on osx
- Air strike 3
- Jo bheji thi dua song
Assuming that energy levels are equally spaced, we use Equation 7.58 to estimate the spacing. These photons are emitted when the molecule makes a transition between two adjacent vibrational energy levels. The lowest-frequency line corresponds to the emission of lowest-frequency photons. What is the spacing between the vibrational energies of this molecule? What is the force constant k of the atomic bond in the HCl molecule? The infrared vibrational spectrum measured for hydrogen chloride has the lowest-frequency line centered at f = 8.88 × 10 13 Hz f = 8.88 × 10 13 Hz. Because the chlorine atom is 35 times more massive than the hydrogen atom, the vibrations of the HCl molecule can be quite well approximated by assuming that the Cl atom is motionless and the H atom performs harmonic oscillations due to an elastic molecular force modeled by Hooke’s law. Vibrational Energies of the Hydrogen Chloride MoleculeThe HCl diatomic molecule consists of one chlorine atom and one hydrogen atom. In this way the potential energy function can be written in a more general form, A first step toward a quantum formulation is to use the classical expression k = m ω 2 k = m ω 2 to limit mention of a “spring” constant between the atoms. We cannot use it, for example, to describe vibrations of diatomic molecules, where quantum effects are important. One problem with this classical formulation is that it is not general. It is a statement about how quickly the object moves through various regions.) The Quantum Harmonic Oscillator (Note that this is not a statement of preference of the object to go to lower energy. Therefore, the probability of finding a classical oscillator between the turning points is highest near the turning points and lowest at the equilibrium position. When an object oscillates, no matter how big or small its energy may be, it spends the longest time near the turning points, because this is where it slows down and reverses its direction of motion. The zero-energy state of a classical oscillator simply means no oscillations and no motion at all (a classical particle sitting at the bottom of the potential well in Figure 7.13). The lowest energy that a classical oscillator may have is zero, which corresponds to a situation where an object is at rest at its equilibrium position. The energy of a classical oscillator changes in a continuous way. Physically, it means that a classical oscillator can never be found beyond its turning points, and its energy depends only on how far the turning points are from its equilibrium position. In this plot, the motion of a classical oscillator is confined to the region where its kinetic energy is nonnegative, which is what the energy relation Equation 7.53 says. The energy of oscillations is E = k A 2 / 2. The position of the object varies periodically in time with angular frequency ω = k / m, ω = k / m, which depends on the mass m of the oscillator and on the force constant k of the net force, and can be written asįigure 7.13 The potential energy well of a classical harmonic oscillator: The motion is confined between turning points at x = − A x = − A and at x = + A x = + A.
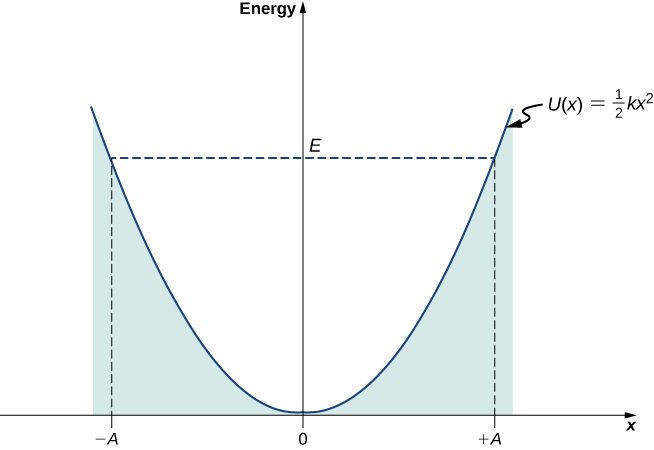
The motion takes place between two turning points, x = ± A x = ± A, where A denotes the amplitude of the motion. In classical mechanics, the particle moves in response to a linear restoring force given by F x = − k x, F x = − k x, where x is the displacement of the particle from its equilibrium position. X-direction about the equilibrium position, x = 0 x = 0. Suppose a mass moves back-and-forth along the In this section, we consider oscillations in one-dimension only. The Classic Harmonic OscillatorĪ simple harmonic oscillator is a particle or system that undergoes harmonic motion about an equilibrium position, such as an object with mass vibrating on a spring. We begin with a review of the classic harmonic oscillator. In this chapter, we begin to study oscillating systems using quantum mechanics. In previous chapters, we used Newtonian mechanics to study macroscopic oscillations, such as a block on a spring and a simple pendulum. Oscillations are found throughout nature, in such things as electromagnetic waves, vibrating molecules, and the gentle back-and-forth sway of a tree branch.
Explain physical situations where the classical and the quantum models coincide.Identify differences between the classical and quantum models of the harmonic oscillator.Describe the model of the quantum harmonic oscillator.By the end of this section, you will be able to:
- Blog
- Will modbus poll work with ab plcs
- Basic lettering lessons tajima pulse
- Nikon scan 4000 ed bit depth
- Winchester model 25 hornet magazine
- Free download parasite in city android
- Harley davidson golf cart parts 50252-74
- Eternal poison cheat engine
- Quantum harmonic oscillator
- Install haproxy on osx
- Air strike 3
- Jo bheji thi dua song
